Calculate standard heat of combustion of ethanol(C2H5OH(I)). Given that A,H (C2H5OH, I) 278 kJ/mol, AH^° (CO2, g)(H20, I) = 286 kJ/mol. 394 kJ and AH

Use the given standard enthalpies of formation (in kJ/mol) to determine the enthalpy of reaction of the following reaction: NH3(g) + 3F2(g)→NF3(g) + 3HF(g) ΔHf^∘(NH3,g) = - 46.2 ; ΔHf^∘(NF3,g) = -
![Given that the CFSE for [Co(H2O)6]2+ is 7360 cm-1, the calculated value of o in KJmol-1 is?(rounded up to onedecimal places)Correct answer is between '-111.0,-108.0'. Can you explain this answer? - EduRev Given that the CFSE for [Co(H2O)6]2+ is 7360 cm-1, the calculated value of o in KJmol-1 is?(rounded up to onedecimal places)Correct answer is between '-111.0,-108.0'. Can you explain this answer? - EduRev](https://edurev.gumlet.io/ApplicationImages/Temp/585d8c36-06da-4fc1-92ad-52ef4e26f9e4_lg.jpg?w=360&dpr=2.6)
Given that the CFSE for [Co(H2O)6]2+ is 7360 cm-1, the calculated value of o in KJmol-1 is?(rounded up to onedecimal places)Correct answer is between '-111.0,-108.0'. Can you explain this answer? - EduRev

SOLVED: PLEASE ANSWER ALL PARTS AND SHOW WORK!!! NEATLY AND EXPLANATION!! Calculate the enthalpy (kJ/mol) of a ternary mixture of ideal gases with the composition 42% CO2, 37% N2, and 21% CO

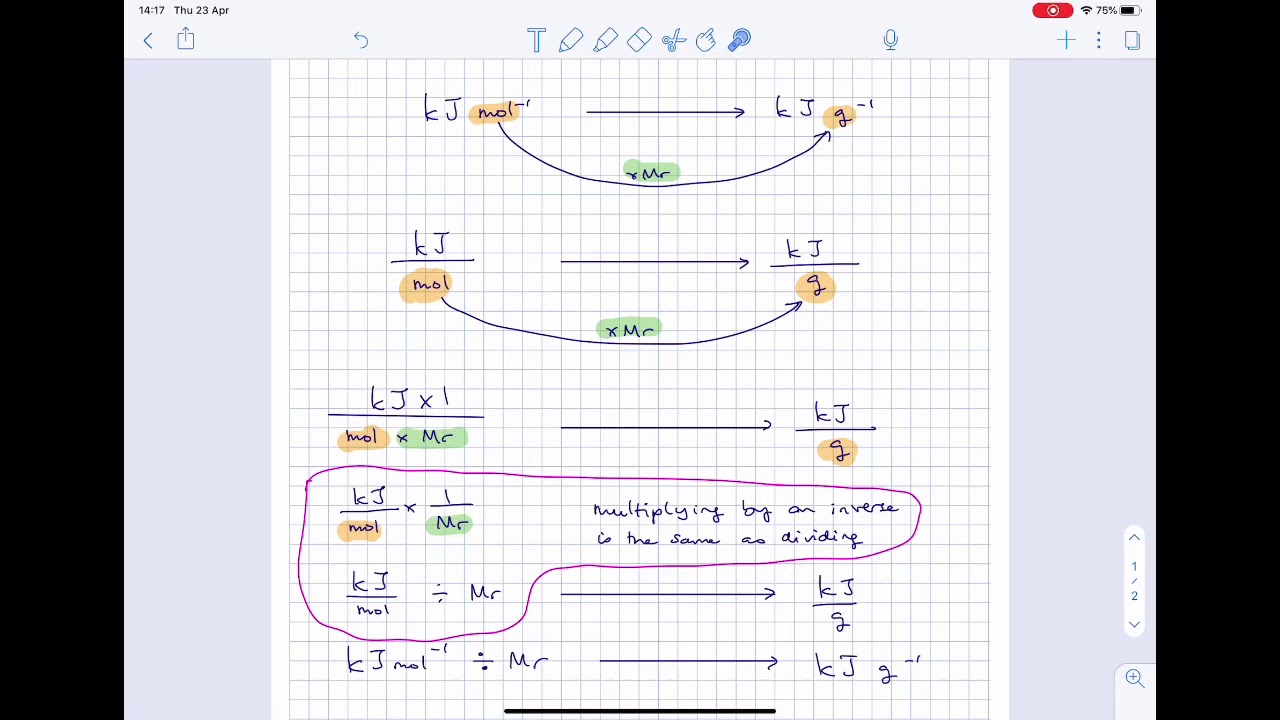
enthalpy - Why is it sometimes kJ only, and in other times kJ/mol? What's the difference? - Chemistry Stack Exchange

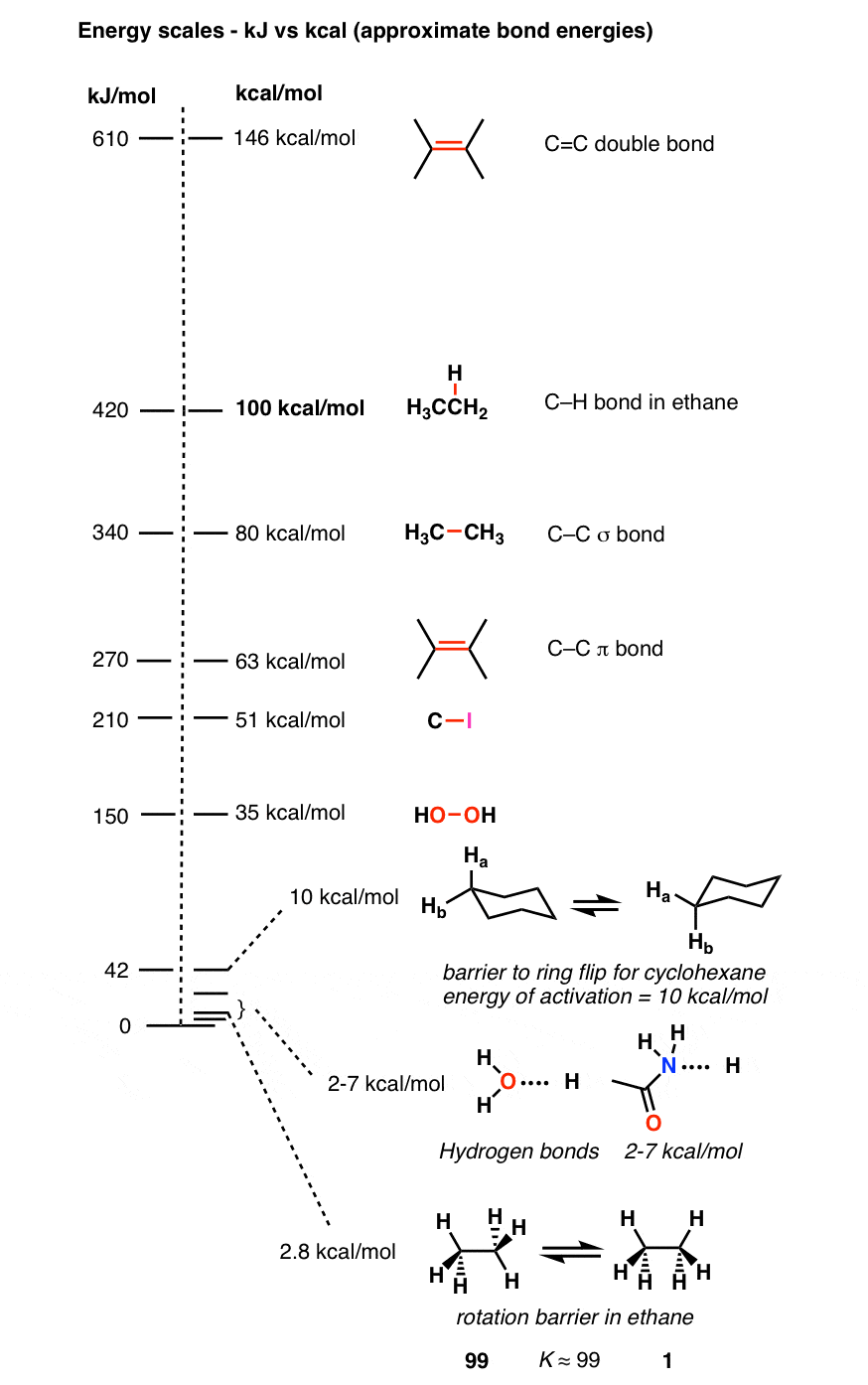
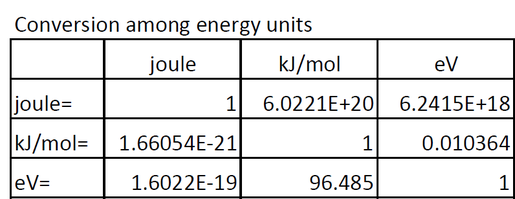
Thermodymanics Lecture 3 8/31/2004. Units Energy Ultimate source of energy is the sun E = h 57 Kcal/mol of photons green light or 238 KJ/mol 1 cal = - ppt download